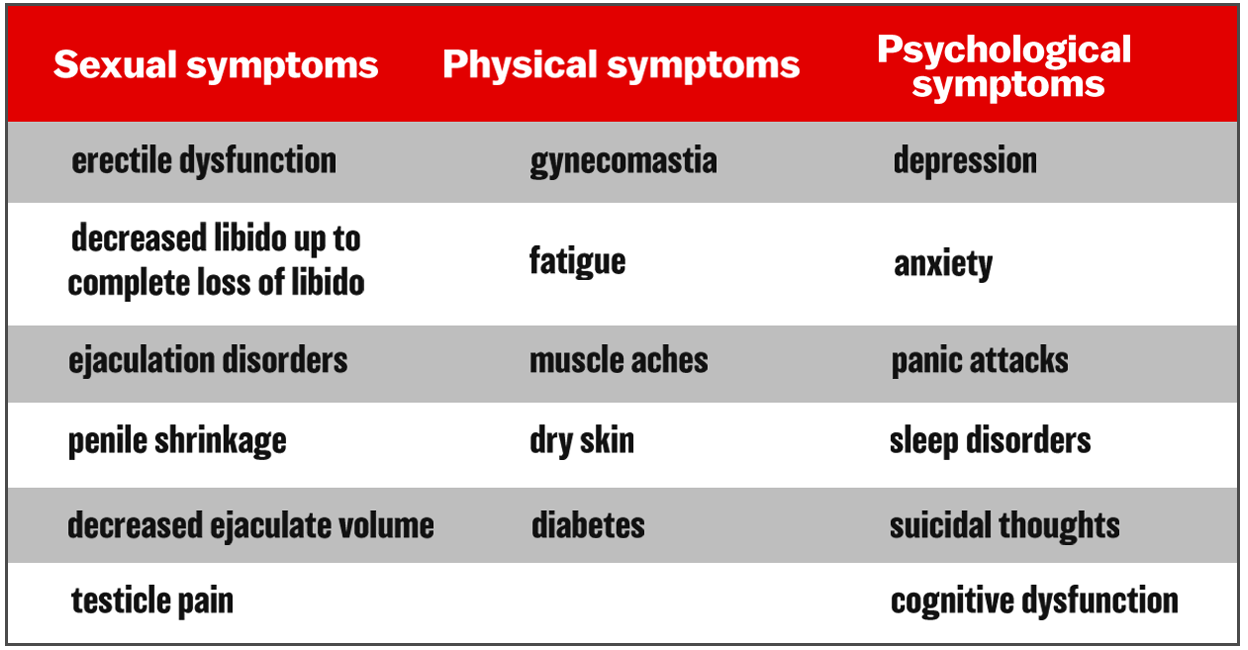
Study Used to Push Non-FDA-Approved Topical Finasteride Knocked by Top German Rx Journal – The Post-Finasteride Syndrome Foundation

Study Used to Push Non-FDA-Approved Topical Finasteride Knocked by Top German Rx Journal – The Post-Finasteride Syndrome Foundation

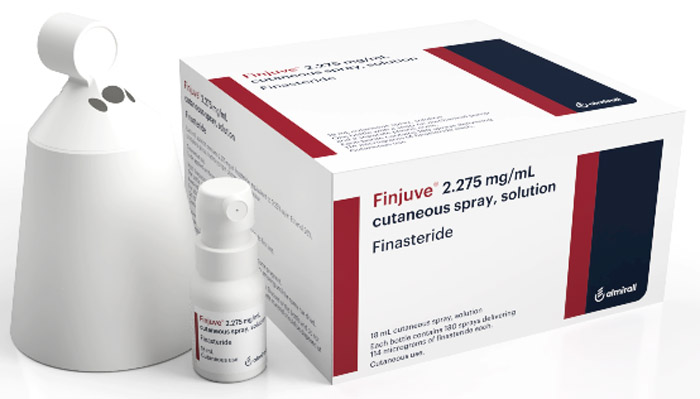
Hikma and Almirall enter an exclusive licensing agreement to market FinjuveTM in the Middle East and North Africa region


![Fuck탈모] 탈모 치료의 혁신이 일어났네요 Fuck탈모] 탈모 치료의 혁신이 일어났네요](https://daedamo.com/new/data/file/story/3543796440_ojGtcexi_1d37b296db599e873bb7dd1c54e7efc4b878cbb2.jpeg)
![핀쥬베 스프레이 [180puf] | 약물정보 | 의료정보 | 건강정보 | 서울아산병원 핀쥬베 스프레이 [180puf] | 약물정보 | 의료정보 | 건강정보 | 서울아산병원](https://heart.amc.seoul.kr/asan/file/viewDomainDrugFile.do?fileName=/P_EA-FINJUV18.jpg)








![Eng] Hair Loss Medication vs. 'Finjuve Spray' | A Dermatologist Talks about Them - YouTube Eng] Hair Loss Medication vs. 'Finjuve Spray' | A Dermatologist Talks about Them - YouTube](https://i.ytimg.com/vi/LiFAzG1FT6Q/maxresdefault.jpg)